
The firm said the instrument uses the same reagents, consumables, and software as the firm's Atellica Solution instrument but is designed for use in low- to medium-volume laboratories, with throughput of up to 1,120 tests per hour. Siemens also gained FDA clearance in July for its Atellica CI Analyzer for immunoassay and clinical chemistry testing. The assay results should be considered preliminary until confirmation through other methods, preferably gas chromatography/mass spectrometry or liquid chromatography/mass spectrometry, the FDA said. The Emit II Plus Buprenorphine Assay, which was designed for use in various chemistry analyzers, is now also cleared for use on Beckman Coulter's DxC 500 AU analyzer. Beckman Coulter said the analyzer uses the same standardized assays and reagents as the firm's other AU clinical chemistry analyzers.Įrlangen, Germany-based Siemens Healthineers also nabbed an FDA clearance for an update to its homogenous enzyme immunoassay used for qualitative and semi-quantitative determination of buprenorphine in urine. The DxC 500 AU Clinical Chemistry Analyzer is used to measure analytes in fluid samples including serum, plasma, and urine. The I-Stat CG8+ cartridge is used for quantification of glucose in whole blood to aid the diagnosis, monitoring, and treatment of carbohydrate metabolism disorders such as diabetes mellitus, neonatal hypoglycemia, idiopathic hypoglycemia, and pancreatic islet cell carcinoma.ĭanaher subsidiary Beckman Coulter secured 510(k) clearance for an automated photometric chemistry analyzer designed for use in small- to medium-volume laboratories along with tests and reagents used for quantitative measurements of glucose, C-reactive protein, sodium, potassium, and chloride. It is designed for use with Abbott's handheld I-Stat 1 analyzer.Ībbott also secured a separate 510(k) clearance in July for a point-of-care glucose test system for the I-Stat 1 System. The test is used to monitor patients receiving anticoagulant therapy with coumarin derivatives, with results reported in seconds and an international normalized ratio.  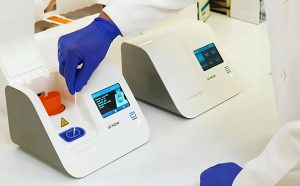
The Abbott Park, Illinois-based firm's I-Stat PTplus Cartridge prothrombin time test is used to quantify the clot time of the extrinsic coagulation pathway when activated by thromboplastin in non-anticoagulated whole blood. The results are used to aid the diagnosis and treatment of liver, bone, parathyroid, and intestinal diseases. The firm's Alkaline Phosphatase2 clinical chemistry assay is used for quantitation of alkaline phosphatase in serum or plasma on Architect C System immunoassay analyzers. NEW YORK – The US Food and Drug Administration in July granted 510(k) clearances for clinical chemistry tests, infectious disease assays, and testing instruments by Abbott Laboratories, Beckman Coulter, Siemens Healthineers, and Roche, among others.Ībbott got the agency's go-ahead for an automated test for alkaline phosphatase and a pair of point-of-care tests for prothrombin time and glucose quantification.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
